Even stronger than a radial tyre? There is every indication that TUBALL carbon nanotubes will revolutionise tyre chemistry even more than silica did in the 1990s and be even more significant than the invention of radial tyres after WWII. Even the tiniest amount of these small tubes, which have a diameter of just 1 nm (one-billionth of a metre) and a wall thickness of one (!) carbon atom, improve tyre characteristics to a mind-blowing extent. The history of the invention is equally remarkable; moreover, it comes from the heart of Siberia.
In 1945, after the nuclear bombs, society realised that matter holds massive amounts of energy, but it is difficult to retrieve it. The necessity to work at an atomic level makes carbon nanotubes extraordinary and, at the same time, difficult to synthesise.

Carbon for Dummies
You cannot examine an advanced technology such as this without the appropriate background, even if you think you know what carbon is. It was probably more than 500 000 years ago when our ancestors began to use charcoal for heating and cooking, but it has been less than three centuries since coal and the steam engine marked the beginning of industrial era. However, neither the prehistoric nor the industrial period of carbon history has anything to do with contemporary nanochemistry..
Broadly speaking, everything growing and living on earth depends on carbon, which is the basis for the chemistry of every living creature. The human body is 65% water and, therefore, is rich in oxygen (65%); carbon ranks second at 18%, then hydrogen at 10%, nitrogen at 3%, and the calcium in our bones accounts for 1.5% of our mass. Nature has more than a million different combinations of carbon and hydrogen, and hydrocarbons are our primary energy source apart from coal; in brief, carbon is irreplaceable and not an element easy to live without.
In its natural state, carbon has only two crystalline forms, which are very different from each other: diamond and graphite. The first is valuable and exceedingly rare and hard, the second is greasy to the touch and far more trivial, with its infirm and pliable appearance; its production reaches about 1.5 million tonnes per year. Few people know that diamond decays over time (a very, very long time!) and degrades to graphite, which ultimately is the most stable form of carbon. Over the centuries, we have become very familiar with this black and grey mineral that has provided us with easy-to-use writing and drawing tools – Chinese ink and pencil lead. Today, on top of everything else, it ensures safety at nuclear power stations and gives us millions of electrical batteries; it is an incontestable ancestor of all other forms of ultraeffective carbon structures that are yet to be created.

From a Micrometre …
Graphite’s useful lubricating properties are provided by the simplicity with which its multiple layers slide against each other; however, chemists are more concerned with its structure. These layers are flat and extremely thin, honeycomb-shaped, and consist of hexagonal rings closely fitted to each other; at the top of each there is a carbon atom connected with three of its neighbours. There are layers only one atom thick, and such a structure facilitates (although relatively) the access to carbon atoms. Although we have been aware of graphite’s enormous potential for a long time, this does not make working at the atomic level any simpler. The main pitfall is that we can only clearly examine these structures once new powerful high-resolution electron microscopes are available.
Starting out from hydrocarbons, chemists first examine carbon in terms of the simplicity with which it converts into fibre. By connecting long and flat microcrystals and aligning them in parallel lines, it is possible to synthesise fibres with a diameter of 5–10 microns. Assembling 1, 3, 6, 12, 24, 48 thousand of such carbon fibres, depending on their intended use, helps synthesise amazingly durable but lightweight threads. In order to restore theit textile industry that had been destroyed by the war, the Japanese began developing carbon fibre in 1959; the first research centre later became Toray Industries, the world leader in the industry.
 Some exceptional features of single wall carbon nanotubes: their conductivity is better than that of copper; at the same time, they are five times lighter than copper, they are 100 times stronger than steel, their length is a million times longer than their diameter, and one gram covers an area of two basketball courts!
Some exceptional features of single wall carbon nanotubes: their conductivity is better than that of copper; at the same time, they are five times lighter than copper, they are 100 times stronger than steel, their length is a million times longer than their diameter, and one gram covers an area of two basketball courts!
These new fibres were not exactly suitable for traditional textiles but, thanks to their mechanical properties, they were rapidly recognised in the military and aircraft industries. Today, the most modern aeroplanes comprise more than 50% carbon fibre, and the A380 could not even fly without it. Everywhere where effectiveness and low weight are required, be it sports equipment, sailing boats, racing cars, or prosthetics, we cannot do without carbon fibre.
… to a Nanometre
However, it was until 1985 when the third crystalline form of carbon was created; this time, it was completely artificial, and it was given the name fullerene. This radically changed the scale of operation: micron-long fibres were replaced with nanometre-long ones, and the dive into the depths of the infinitely small began. The prefix nano - ( nanos in Greek) means a one-billionth part (of a metre). When you play with atoms on a nanoscale, you have to divide measurements in microns by 1000! The discovery of fullerenes happened in a laboratory while attempting to answer astrophysicists’ questions about how some long carbon-containing chains that had been discovered in space were formed.
Using the two-dimensional layers of graphite, chemists created new three-dimensional molecules that, just as before, consisted of 100% carbon but could take on various more interesting forms, such as spheres, ellipsoids, tubes and rings. The method used was evaporation in the neutral medium of a graphite disc by laser ablation under very specific conditions. The idea itself, let alone its implementation, is extraordinary – and this was officially recognised in 1996 when Richard E. Smalley, Robert F. Curl Jr and Sir Harold W. Kroto, an Anglo-American team of inventors, received the Nobel Prize in Chemistry. That was only fair.
The very first product was shaped like a soccer ball. Just like a ball, it was composed of 20 hexagons connected with 12 pentagons as in graphite; however, the new structure, named C 60 , was only 0.7 nanometres thick with an internal diameter of 1 nanometre, which is 200 million times less than a real soccer ball! The research team was quite imaginative when naming the new product. For a while, C 60 was called the bucky-ball after Buckminster Fuller, the inventor of geodesic spheres, then it became first buckminsterfullerene, and later was shortened (luckily!) to fullerene.
Smalley, Curl and Kroto put their foot in the door and kick-started the process: numerous other research teams then rushed to obtain fullerenes, developing other methods of synthesis. The structures became more diverse, more effective, with their qualities being both different and outstanding! It is now believed there are more than 250 000 types of fullerenes (and the number is still increasing) that can be applied in any industry, be it pharmaceuticals, cosmetics, electronics, photogalvanics, lubricants, etc. Nanoparticles are the things most used in the world – apart from money.
Enter Nanotubes and Graphene
After C 60 , “soccer balls” containing 70, 76, 84, 100 and 200, and even 20, carbon atoms were produced, and that was only the beginning. Carbon molecules divide when exposed to temperature (you just have to learn how to do this), and their atoms recombine in infinitely multiple forms so that any configuration seems possible. With balls, megatubes, nanotubes, dimers, polymers, nano-onions, etc., the fullerene family is ever-growing, but the industry’s greatest hopes still lie with tiny nanotubes.
Carbon fibre and fullerenes were born in 1959 and 1985 respectively; nanotubes appeared between 1991 and 1993. In 1991, Sumio Iijima, a pioneering researcher from the Japanese company NEC, was studying fullerene synthesis and produced the first multiwall nanotubes with the number of graphene layers varying from two to 50. He was able to produce them again in 1993, but this time they were single wall nanotubes. Donald S. Bethune from IBM did the same at the same time, but in his own unique way.
This stage of carbon history is notable for the appearance of a new material that forms single wall nanotube walls, and that is graphene. It is a famous two-dimensional crystal with a flat honeycomb-shaped layer that is only one atom thick; it is its layers that form graphite. In reality, working with a material that seemed so simple because of its natural origin turned out not to be simple at all. It was not until 2004 when the Soviet-born Dutch-British physicist André Geim was able to obtain this one atom thick film in a very unusual way. He used an adhesive tape to remove the substance layer by layer until he produced a famous single-atom layer. Of course, other methods of graphene production were invented as well, but it was this one that brought the Nobel Prize to Geim in 2010. He shared the prize with Konstantin Novoselov, a UK national born in Russia, who also now works in the UK.
According to conventional wisdom, graphene will revolutionise our life; some even believe that it is a technology breakthrough comparable to the transition from the Bronze Age to the Iron Age! Graphene is both flexible and elastic, it conducts electricity better than copper, it is 100–300 times stronger and six times lighter than steel and it has no colour. This unique material is suitable for almost any use and can reinforce almost anything despite its size: it is one million times as thin as a hair, and three million layers of graphene together are no thicker than 1 mm. The entire world led by Europe spends billions to synthesise properly sized graphene layers at an acceptable price, but not all succeed!
Single Wall Carbon Nanotubes
While commercial synthesis of graphene has yet to be launched, another form of fullerene with graphene walls is gaining momentum. It is a nanotube. Initially, Iijima produced it using two graphite electrodes, with an electric current generating a 6000°C plasma: the positive anode evaporates and the negative cathode acquires a blackish deposit which is nanotubes. There are other methods apart from this arc-discharge plasma diffusion. They require high or medium temperatures or a gaseous state, but the results are different, although carbon atoms, once released, enthusiastically recombine creating fancy forms. Therefore, most of the synthesised nanotubes, coming from the fullerene family, are “closed” at their edges with one or two semi-spherical caps. These “soccer ball halves” can be kept or removed to open the tube from both sides and fill it with other products to make its features more appealing.
Multiwall carbon nanotubes sometimes resemble Russian nested dolls: there are many tubes with decreasing diameters coiled inside each other, or one coiled scroll-like layer. Other structures can have gaps or holes, be honeycomb-shaped or have five or seven sides; sometimes they contain impurities, deposits from a metal catalytic agent, which are indispensable for this operation. These nanotubes must be cleaned or restored before use. Single wall carbon nanotubes (SWCNT) can also have very different structures (including spirals), which gives them better mechanical and electrical parameters, or conductor or semi-conductor properties, etc.
Whatever the method might be, the development of nanotube synthesis is not an easy task; it is an extremely challenging process where very small quantities are obtained at a high cost. There are still many challenges and it is hard to avoid them, as it turned out in 2013, when Bayer, a large chemical corporation, left the game and lost a lot of money closing its plant in Leverkusen with a synthesis capacity of 200 tonnes of nanotubes per year just three years after its start-up. It seems that this decision was prompted by technical (with carbon fibre and kevlar still being used) and commercial competition as well as overestimated demand for volume and growth rates.
OCSiAl, Baby of the Silicon Taiga
Similar to many other inventions with multiple authorship, nanotubes were discovered not only by Iijima and Bethune. Many teams worked in this area, sometimes without even knowing about each other and using different methods. A closer study of the background shows that the Soviet scientists Radushkevich and Lukyanovich observed 50 nanometre tubes as early as 1952, while, in 1976, Oberlin, Endo and Koyama observed hollow fibres and SWNCT. In 1979, John Abrahamson confirmed and, in 1981, Soviet scientists produced an image of coiling graphene, SWCNT within a range between 0.6 and 6 nanometres.
The Cold War and protection of technological secrets slowed down the distribution of this helpful information, which is how OCSiAl, a Russian company based in Akademgorodok, a research town 20 km from Novosibirsk in the heart of Siberia, came to the global market. Akademgorodok was inspired and set up by the academician Mikhail Lavrentyev, a doctor in physics and mathematics, in 1957. Nikita Khruschev indulged him to create the best working and living conditions for the Soviet science elite. The research town functioned fairly well but it was abandoned after the demise of the Soviet Union. Fortunately, it was later revived in a new, updated, entrepreneurial economy. This community with 60 000 residents is currently the cradle of world-class start-ups. A new science park opened there in 2006. This dynamics and creative community, with a high concentration of advanced technologies, has led to Akademgorodok being called the Silicon Taiga, by analogy with the Californian Silicon Valley.
OCSiAl, the name of the company, follows the best scientific traditions and refers to the chemical symbols of the main elements the company handles: O stands for oxygen, C for carbon (with its atomic number of 6 included in the logo), Si for silicon and Al for aluminium.
The Three Musketeers of OCSiAl
Just like in Dumas’s story, the three musketeers who founded OCSiAl were, in fact, four people! Although officially Mikhail Predtechensky is only Senior Vice President and author of the synthesis technology, he is the key figure and the man of the future. He is the scientist and inventor who was able to refine the plasmochemical reactor that is capable of synthesising large quantities of the highest quality SWCNT at market prices, being the first in this respect. Having this advanced technology, he was joined by three other co-founders. These equally outstanding finance and management experts are Yuri Koropachinsky, Oleg Kirilov and Yuri Zelvensky, the latter who now lives in Israel. They managed to determine the global market potential (estimated at USD 3 billion!) and to raise the USD 350 million required to set up OCSiAl in 2009, and they registered patents in 2013 and built the Graphetron 1.0 reactor capable of synthesising 10 tonnes of SWCNT per year.
 Graphetron 1.0 was launched in 2014 and, by 2016, the company
employed 260 people with 100 top scientists working in Akademgorodok
laboratories. Other employees include engineers and salespeople who sell
branded TUBALL nanotubes all over the world. Initially, the company opened
offices in Columbus, Sunnyvale, Incheon, Mumbai, Shenzhen, Hong Kong and Moscow
to get access to the largest markets, and the most recent office, the
headquarters, has now been located in Luxembourg for several months. It goes
without saying that this has been a massive and persistent effort supported by
expert teams working in different areas since there are many diverse industries
whose products can be enhanced by TUBALL. Engineers and salespeople are
confident about the quality and wide application of TUBALL, which is very good
as the OCSiAl marketing department sets very challenging targets. The second
reactor, which will be capable of synthesising 50 tonnes per year, is
planned to be started in 2017. Short-term forecasts grow exponentially, based
on 800 tonnes in 2020 and 3000 tonnes in 2022.
Graphetron 1.0 was launched in 2014 and, by 2016, the company
employed 260 people with 100 top scientists working in Akademgorodok
laboratories. Other employees include engineers and salespeople who sell
branded TUBALL nanotubes all over the world. Initially, the company opened
offices in Columbus, Sunnyvale, Incheon, Mumbai, Shenzhen, Hong Kong and Moscow
to get access to the largest markets, and the most recent office, the
headquarters, has now been located in Luxembourg for several months. It goes
without saying that this has been a massive and persistent effort supported by
expert teams working in different areas since there are many diverse industries
whose products can be enhanced by TUBALL. Engineers and salespeople are
confident about the quality and wide application of TUBALL, which is very good
as the OCSiAl marketing department sets very challenging targets. The second
reactor, which will be capable of synthesising 50 tonnes per year, is
planned to be started in 2017. Short-term forecasts grow exponentially, based
on 800 tonnes in 2020 and 3000 tonnes in 2022.
The first two graphetrons will start synthesising 60 tonnes each in Akademgorodok in 2018, but the third one is expected to be installed closer to Europe and its main markets. Since the main specifications require “a lot of energy and gas”, bets are being placed as to its future location. It might as well be Luxembourg, with the company headquarters located there, and the Grand Duchy having to get rid of everything “bank-related”.
Obvious Excellence
Such forecasts could be considered too optimistic – shouldn’t we remember the example of Bayer? However, there are no such fears in Luxembourg, as TUBALL SWCNT is so much better than multiwall nanotubes. This is the adamant belief of Cristoph Siara, Sales and Marketing Director at OCSiAl Europe, and of Jean-Nicolas Helt, Development and Support Leader for Elastomers at OCSiAl Europe. Judging by his name, you would not say that Cristoph Siara is German. Cristoph had a legal education. He has lived in France since 1983. The transfer from one advanced area to the other helped him expertly master the most complex technologies. When he speaks about nanotubes, you would think he is a chemist. Engineer Jean-Nicolas Helt was born in France. He obtained a degree in physics from Nancy University and then from ESEM [1] in Orléans. His excellent education enabled him to conduct in-depth studies at Goodyear in Luxembourg. He can boast of several fundamental achievements in the tyre industry made over his 17 years in the heavy truck and car sector. He joined OCSiAl in 2015 as a project manager and it was he who said that TUBALL could bring something valuable to the tyre industry.
Cristoph Siara explains that TUBALL SWCNT is, first of all, a large breakthrough as compared with its predecessor multiwall nanotubes that were marketed before. With a diameter of 25–40 nm, being short with several twisted layers, these multiwall nanotubes were rigid, which significantly impaired their mechanical properties and involved health issues. Even if carbon (inert) is neutral to the human body, its behaviour is comparable to that of asbestos fibres that attach to cells. However, this can be easily avoided with thin and flexible TUBALL products. In reality, unlike multilayer nanotubes, TUBALL SWCNTs are thin (about 1.5 nm) and very long (over 5 micron). their length is 3000 times their width, which becomes clear in an example from another scale: they are like a 100 metre long garden hose!
It means we have a linguistic problem here as such words as “ticker tape”, “noodles” or “hollow and long carbon fibre” seem more suitable than “tube”. But it is so much easier to use the word “nanotube”!
Here are some other aspects where TUBALL remains unrivalled: its 1 nm layer is absolutely even, the amorphous carbon content is less than 10%, and the residual non-organic impurities (Fer) are below 15% and encapsulated, i.e. inactive. Unlike their competitors, TUBALL products do not require purification or functionalisation. Other characteristics worth mentioning are a carbon content of over 85% and a G/D band ratio (Raman spectrometry) of over 70, which confirms excellent conductivity. All these findings have been confirmed by independent laboratories, including Intertek (May 2014).

Standard example of a quantum leap in performance of a leakproof synthetic nitrile rubber seal: incredible growth of all parameters.
Process Makes the Difference
Mikhail Predtechensky’s graphetron is probably one of the devices that will revolutionise the 21st century. OCSiAl teaches us that it is about a continuous process capable of processing large volumes using precursors and an inexpensive catalytic agent, probably iron-based, judging from residual admixtures. How does it work? That is an absolute and well-guarded secret. Cristoph Siara and Jean-Nicolas Helt laughed and said that they know nothing about it. The first pre-employment document they signed, similarly to all other employees, was a non-disclosure agreement! Graphetron V1.0 was expected to be shown at a scientific conference in November, but the secret was not unveiled. The most important thing about it is that it generates a continuous flow of high-quality SWCNT at affordable prices. These 10 tonnes annually currently account for 90% of the global synthesis of SWCNT, and 50 tonnes more will be synthesised in Novosibirsk from 2017 onwards!
How are TUBALL products priced? This also is forbidden ground, a trade secret. You can get some idea from OCSiAl sales materials, but you can’t help feeling that these figures are very far from true estimates. Delivery from Novosibirsk will cost you USD 8 per gram for small orders and USD 2 per gram for large ones. OCSiAl diffidently assures you that the price has been reduced at least 25 times.
This frantic race to increase tonnage is explained by TUBALL’s multifunctionality. OCSiAl is selling an extraordinarily versatile additive, an amplifier capable of supporting an explosive growth of performance for about 70% of useful materials on the planet, so this is about much more than just carbon nanotubes.
Versatile Additive, Incredible Performance
Mentioning TUBALL’s properties is like doing the splits: the further you go, the better your performance! Let’s be brief. TUBALL remains heat-resistant at up to 1000°C, it is 100 times stronger than steel, and its area is so large that it cannot be reasonably perceived: 1 gram of TUBALL is enough to cover two basketball courts, that is, 3000 m 2 . Finally, it is the best electricity conductor available, and it is six times lighter than copper.
All this would be of little use without a fundamental property, which is its amazing capability to dissipate. Very thin and long TUBALL tubes create numerous grids and incredible agglomerates that inconspicuously mix with other elements, making them stronger. Therefore, a minor amount of TUBALL – from 1/1000 to 1/10 000 of the total weight – is enough to trigger an explosive growth of performance. An SWCNT is the real SOLUTION for a large number of technological breakthroughs in the 21st century.
A small bottle with 1 gram of TUBALL that OCSiAl visitors can hold to better “feel” the product is a 100% guarantee of the result when the details of its contents are disclosed. That bottle contains 10 15 pieces, i.e. 1 000 000 000 000 000 (one million billion) tubes! If you place them back to back, the length will be about 50 million kilometres!
Everything TUBALL is capable of is summarised by OCSiAl in one diagram showing a beautiful flower with multiple petals. When you choose its properties – conductivity, strength, chemical neutrality, clarity, etc. – or add them up, you open up more and more potential applications. TUBALL is truly the “versatile amplifier” that it claims to be.
To simplify its use, OCSiAl rarely supplies it as powder and without supporting applications. It is offered in more convenient application forms: as a liquid, polymer, oil, rubber, etc. and even as a suspension in solvents. This makes its mixing and dispersing much easier. For example, 50 g of TUBALL dissolved in 50 kg of epoxy resin or polyester will immediately make these substances conductive, which is a very practical solution for floors – and you can even make them coloured!
Flexibility Means Safety
Such compositions have another advantage: safety during nanotube use. The tubes’ primary form and minute size suggest that they would be able to enter the very core of human cells, and therefore precautions should be taken, even though carbon is not toxic for humans. These smallest particles introduced into the matrix to facilitate their injection into a stimulated material cannot evaporate into the air, which makes them safe to use and puts at ease those who fear potential carcinogenic effects, as is the case with asbestos. The World Health Organization (WHO) assumes that nanotubes resemble fibres, but TUBALL SWCNT behaviour is very different from that of the multiwall nanotubes reviewed earlier. “For you to understand it all,” says Cristoph Siara, “if a multiwall nanotube is a golf club, TUBALL is a garden hose. Multiwall nanotubes are solid and rough, and they can enter cells and attach to them causing problems. This is not the case with long flexible TUBALL nanotubes that do not penetrate the cell.”
OCSiAl treats this aspect with great care and monitors all studies being undertaken in Europe and around the world. Since 2008, it is collaborating with BAuA, a German government institution involved in developing industrial regulations and in particular determining product characteristics for employee safety. TUBALL was analysed in its simplest powder form (purchased by 10% of customers), and its health effects were considered favourable. Only one issue remains: all attempts to remove nanotubes from the air by filtration fail as their small size makes them evade all known filter materials! While the solution is being developed (the work is under way), OCSiAl follows all precautions and suggests that the most effective safeguards mandatory for handling dangerous chemicals are used when working with TUBALL powder. These include a full-face mask, overalls, gloves and boots. Goggles, gloves and overalls are sufficient protection when handling liquids.
OCSiAl takes care of their products’ integral life cycle. The news is comforting, because after they are implemented into the matrix and then in the new materials, nanotubes stay put there. With all necessary protection against potential hazards, TUBALL nanotubes become a “regular” chemical that meets the most stringent recent recommendations. Therefore, in October, OCSiAl gratefully received a Reach Certificate for 10 tonnes of nanotubes, although it was hardly a surprise.
The Great Tyre Revolution
Since rubber tyres were first invented, there have been increasing discussions about reinforcing fillers. From clay to talc and then to carbon, we have been trying to improve tyre strength. But in 1991, silica changed the entire situation. It makes rubber versatile and capable of adapting to specific loads. Silica became integral to tyre efficiency. Nevertheless, the impact of silica will be insignificant when compared with the sharp leap expected once TUBALL comes to the tyre industry.
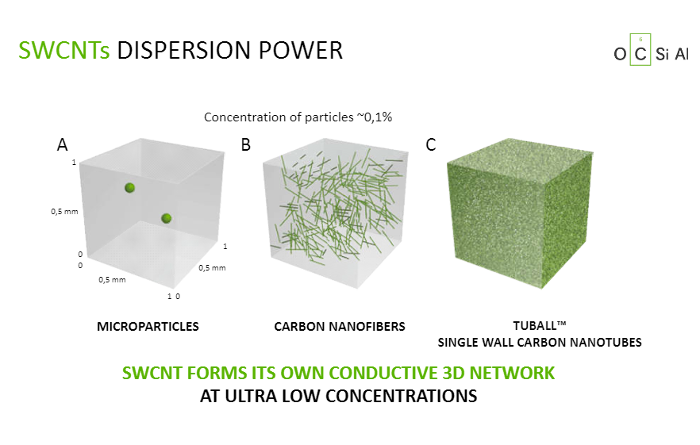
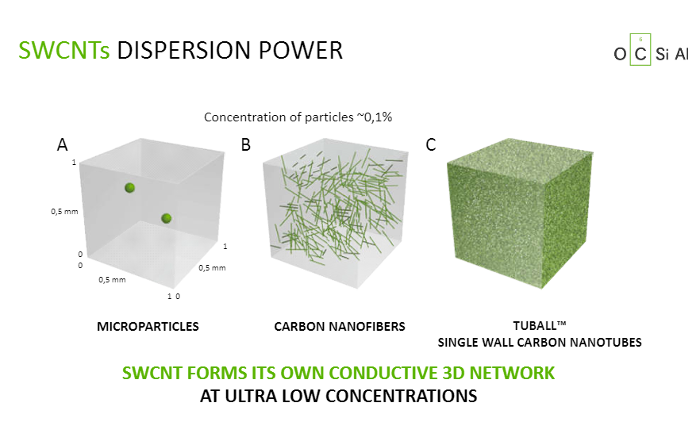
After 17 years of work at Goodyear, Jean-Nicolas Helt is adamantly pursuing the goal. The diagram demonstrates the incredible dispersion of TUBALL in mixtures designed for tyre manufacturing. There are two black particles of carbon to the left; they look fairly isolated in the polymeric cube. In the centre, there is an attempt to reinforce the product with multiwall carbon nanotubes. They are fairly short, hard and agglomerated, and the reinforcement they provide is very weak and ineffective. To the right, TUBALL in a proportion of only 1/1000 of the total weight fills 100% of the cube’s volume with a very compact grid of intensively intertwined SWCNT. Therefore, this mini-filler has a large reinforcing effect because it is a high-structure substance that increases the connectedness of components. Such reinforced connections are more effective because they reduce the mobility of components and, therefore, their wear and tear. It is quite logical that a 3D SWCNT grid forms a second skeleton within tyre rubber, which slows down ageing. In addition, TUBALL is chemically neutral and therefore more resistant to heat and UV radiation, and doesn’t contribute to hydrocarbon pollution, as compared with the current components.
“Take care,” Jean-Nicolas Helt comments, “TUBALL does not suppress carbon black or silica. The chemical and basic properties of a tyre stay the same; small quantities of SWCNT merely activate them. Another TUBALL advantage is that it is an extremely potent conductor so that a tyre case can be 100% silica and have 100% static electricity conductivity rather than insulating properties. This will make the NdC rubber strip across the case boundary that is currently used in premium-class tyres to ground static electricity redundant. This is yet another benefit of mixing and extrusion.
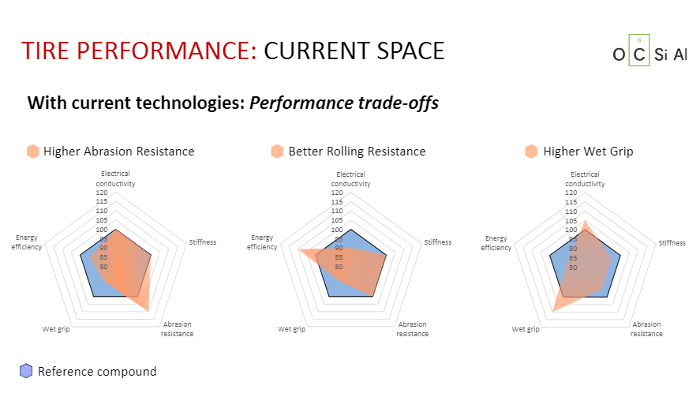
Diagram A. Blue spiders are classical mixture indicators, pink areas demonstrate a benefit obtained with adding silicone. Diagrams to be compared to the next Diagram B addressing the issue with TUBALL added.
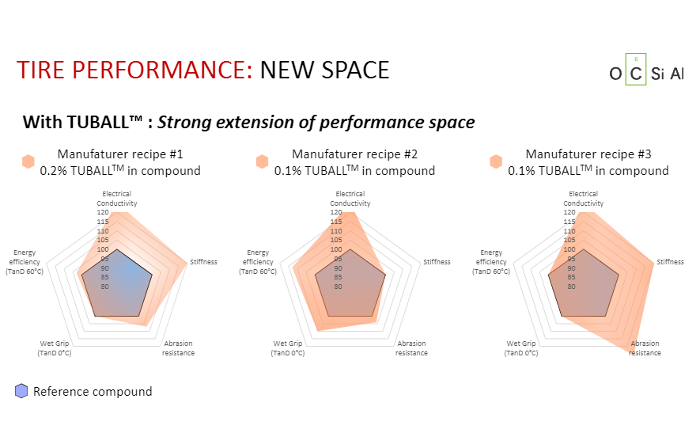
Diagram B. The same principle as is shown in the previous Diagram A with the same scale. It can be concluded that pink surfaces demonstrating improved performance are much longer when TUBALL is added.
Polymer s with TUBALL Additive
TUBALL impacts polymers and reinforcing fillers in a similar way. Because of this, engineers can easily develop à la carte tyres, adding a specific polymer and keeping a specific characteristic which will be in no way affected by development of other indicators. For instance, TUBALL can compensate for the deficiencies of some tyres on dry or wet surfaces. It will do this for motorcycle tyres, too, since there is the potential to improve both traction and wear properties. “It can improve anything,” Jean-Nicolas Helt sums up. But what is the price? Taking into account the small quantity of TUBALL that needs to be added to the mixture (a few thousandths of the total weight) and its reasonable cost, Jean-Nicolas assumes that the manufacturing cost will increase from USD 2 to USD 3 per tyre, which is a substantial but an acceptable increase for premium tyres that will be first to introduce TUBALL since their primary goal is improved performance. And it is absolutely true because many manufacturers are already considering TUBALL, especially after its very convincing tests conducted by independent laboratories such as Smithers, the leading laboratory in the world. It was the time when all OCSiAl claims were put to test, including the one that any amount above the small quantities prescribed does not bring further improvement. Indeed, the conclusion was that you do not have to add more!
The summary further says that TUBALL can be dosed into mixtures very easily since the process (mixing, extrusion, processing, etc.) does not change. You simply need to open a TUBALL tank to transfer the contents to a Bunbury mixer, because OCSiAl supplies TUBALL MATRIX 603 as a concentrate where nanotubes are mixed with synthesised rubber (natural rubber, styrene, nitrile rubber, etc.) and TDAE type processing oil, which is generally used in tyre treads. TUBALL is also available as a suspension with various solvents (MEK, isopropanol, ethylene glycol, ethyl acetate, N-methylpyrrolidone, glycerine and even water). As well as being safe, these ingredients are extremely easy to use.
Easy and ideal for use, this application may be simplified even further with TUBALL added to the polymer during polymerisation – and then you will not need additional operations when mixing! This method of injection at the time of polymer “birth” shifts the problem from the manufacturer to the supplier of synthesised rubber, but OCSiAl has considered this by initiating cooperation with LANXESS. In other words, TUBALL is ready to enter the tyre industry through two doors at the same time and it means that the situation will change faster.
Even if natural rubber can be added only at the mixing stage, TUBALL will still have excellent opportunities of being added to other synthesised rubbers, isoprene or nitrile butadiene directly during manufacturing. Nitrile butadiene rubber has taken linings to new heights of strength in all areas. In other words, the tyre and industrial rubber market (TUBALL is already being used in surgeons’ latex gloves), polymers, elastomers, composites, batteries, photovoltaic arrays, flexible displays, magnetic ink, anti-static concrete, paints, ceramics, copper, semiconductors, window glass, adhesive tapes, etc. are all target areas for TUBALL use. Now we can better understand the Graphetron 50 project that is aimed at achieving an explosive 70% growth of the performance of current industry products.
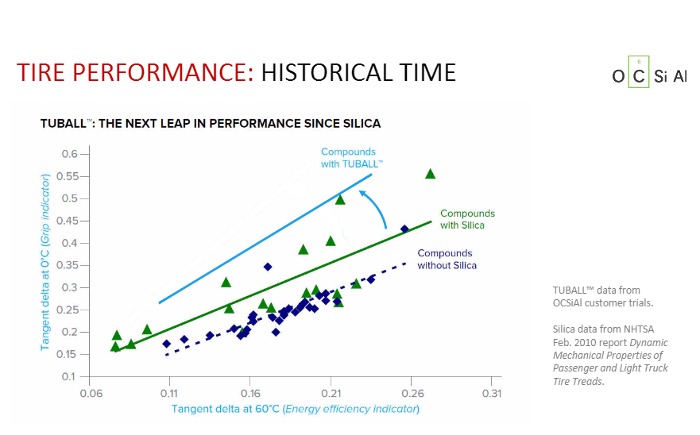
Diagram C. The solid straight line at the bottom shows classical mixtures, the green dashed line shows mixtures with silicone, and the blue cross line shows the performance improvement when TUBALL is added to the mixture.
Competition Has Started
Those who doubt the powerful technological advance of TUBALL in the tyre industry can look at three diagrams by Jean-Nicolas Helt. The first two are spider diagrams where classical tyre performance is compared (three to three) with that of black tyres with silica and that of tyres with TUBALL. Diagram A demonstrates, against a blue octagon, performance of the “black mixture”. The breakthrough achieved thanks to silica is shown in light pink. This is impressive but far from impacting the entire tyre performance range.
Diagram B is based on the same principle but, this time, light pink TUBALL zones fill an incredible area demonstrating a huge increase in performance in almost all sectors. Furthermore, the low percentage of material used (0.2% for natural rubber concentrate and 0.1% for oil concentrates) is truly amazing.
Diagram C has long been known in industry media. Two straight lines excellently demonstrate the “soot mixture” performance (below in dark blue) and the “silica” performance, which is better and is shown in green dashed lines. OCSiAl has added the third line to these tests: lines for TUBALL mixtures are shown above in cyan. This visually represents decisive advantages of SWCNT and, if manufacturers are honest, they must clearly say, as with silica, that improved product performance is achieved with nanotubes.
Some manufacturers are ready to mention the use of nanocarbon, but others might use it too – without saying so. Since the beginning of the year, Vittoria, a bicycle tyre manufacturer, has sold tyres with graphene, the basic material for TUBALL nanotubes. Vittoria uses graphene as layers inserted into the tyre case and claims that it has found a previously unachieved compromise: simultaneous improvement of rolling resistance, lightness, and puncture resistance that is so important for bicycles. Competition confirms Jean-Nicolas Helt’s words about improving many characteristics at once.
The second piece of news is from China where an agreement was signed by Sentury Tire and Huago in August to produce tyres with graphene. We still do not know the details, but the technology will certainly differ from that in Vittoria tyres, which are only 25 mm wide. Such news indicates the overall progress: rolling resistance and mileage multiplied by 1.5. Two major players have already shown their graphene “first-born” at GrapChina, large expert meeting on carbon held on 22 September. At that meeting, Shangdong officially announced that they would manufacture tyres with graphene. All the manufacturers who use it refer to its discovery by the Nobel Prize winners. It is an argument in a dispute that TUBALL cannot challenge even though nanotubes were invented before graphene!
We bet that the number of such news stories will grow very fast. 2016 is a starting point for carbon use in the tyre industry. The shift has just started, and OCSiAl nanotubes are at the forefront of this transformation. This process is worth following – over many years to come.
Jean - Pierre Goss e lin
